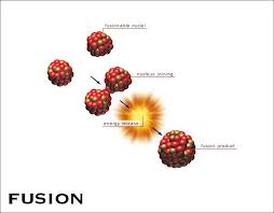
If instead you fuse very light nuclei to get bigger products, energy is again released because the nucleons in the products are more tightly bound than in the original nuclei. Thus, if you split a nucleus that is much larger than iron into smaller fragments, you will release energy because the smaller fragments are at a lower energy than the original nucleus. It turns out that the most tightly bound atomic nuclei are around the size of iron-56. If a nuclear reaction produces nuclei that are more tightly bound than the originals, then the excess energy will be released. The key is in how tightly the nucleons are held together in a nucleus. Nuclear fission is a process in which a nucleus splits into two smaller nuclei. Nuclear fusion is a process in which two nuclei join to form a larger nucleus. This is derived from the term 'anion', used in inorganic chemistry, for a negative ion or an ion that is attracted to the anode or positive electrode.Fusion and fission are similar in that they both release large amounts of energy. Such negative ions are known as carbanions. Heterolytic fission is more likely to happen in when the electrons are shared unequally between the atoms involved in the fission.ĭepending on the nature and electronegativity of the atom attached in place of bromine, the bond can break heterolytically to form a negative ion instead of a carbocation. The table tennis balls represent neutrons that are released when the nucleus splits and cause other nuclei to split. A neutron should be expressed as 1/0 n, not 0/1n. The number of freely-moving neutrons increases over time. 
This is derived from the term 'cation', used in inorganic chemistry, for a positive ion or an ion that is attracted to the cathode or negative electrode. I dont even know what 'Modeling Fission' means. The carbocation (or carbonium ion) is a reactive and therefore, short-lived unit. The compound bromomethane (and others) can split heterolytically as shown: The following are real examples of homolytic fission. Fission is a nuclear reaction in which an atomic nucleus breaks into smaller nuclei of comparable mass, releasing a large amount of energy. The electrons have been re-distributed equally and this is known as homolytic fission. The dots in the equation represent unpaired electrons that came from the covalent bond. The chlorine atom takes both bonding electrons and becomes a negative ion which can also be shown as :Cl - Consider the fission of a single covalent bond in the substance X-Y. This can happen in one of two ways, depending on the electronegativity values of the two atoms concerned:Ī real example of heterolytic fission is shown below to hydrogen chloride. 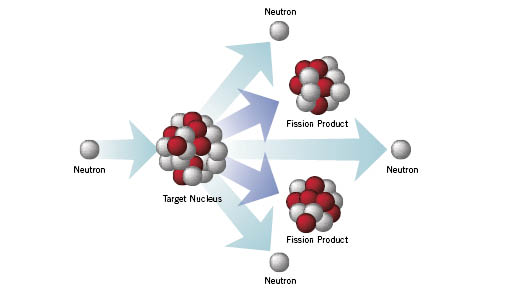
Homolytic fission is more likely to happen in when the electrons are shared equally between the atoms involved in the fission. is also a reactive unit as it has an unpaired electron, but it cannot be described as an atom. This can also happen to bonds in certain organic molecules where R represents part of the organic molecule. As the atoms each have an unpaired electron, they are very reactive units. Two identical bromine atoms are produced from a bromine molecule. The following are real examples of homolytic fission. Consider the fission of a single covalent bond in the substance X-Y X-Y X.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
